Free Newsletter Sign-up!
The Defender®
CHD.TV

Help Us Fight For Legal Justice
Our extensive legal resources raise awareness of our legally protected rights.
When health freedom is under attack, our goal is to encourage, engage and empower the preserve fundamental and human rights.
The greatest crisis that America faces today is the chronic disease epidemic in America’s children.
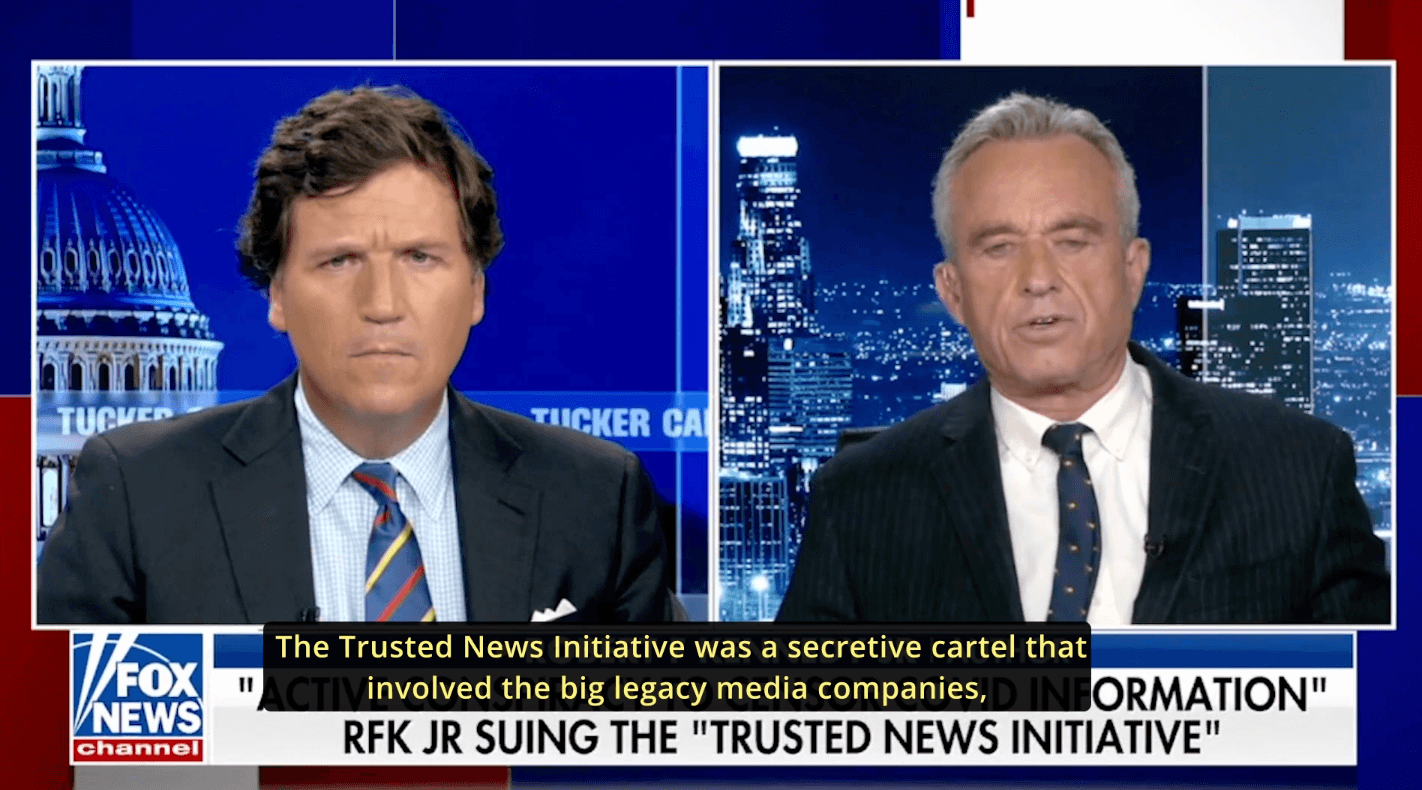
Chairman On Leave
54% of US Youth are Chronically Ill
America's children are facing unprecedented epidemics!
We are in a crisis